- Infarct Size & Inflammation
- Traumatic Brain Injury
- Hypertensive Rat Brain
- Encephalomyelitis
- Cardiovascular Angiogenesis
Juan Carlos Peña-Philippides, Yirong Yang, Olga Bragina, Sean Hagberg, Edwin M. Nemoto, Tamara Roitbak
Published 2014 in Translational Stroke Research – DOI: 10.1007/s12975-014-0334-1
Abstract
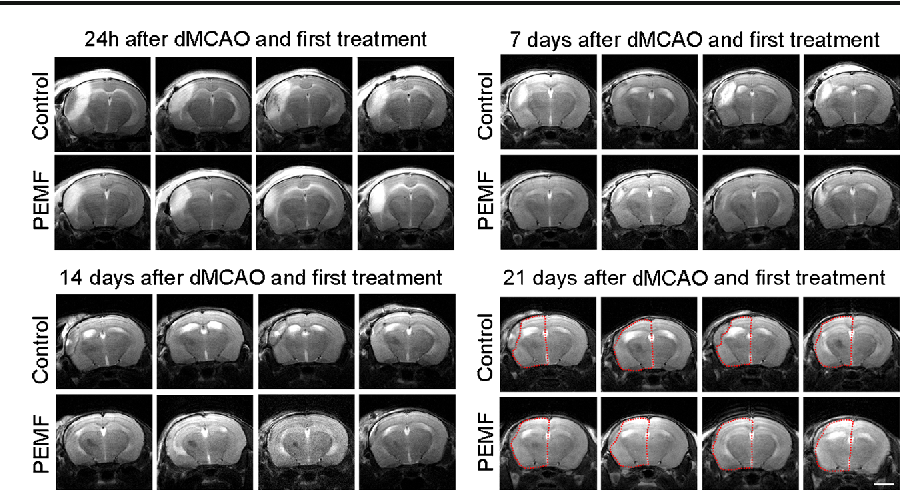
Pulsed electromagnetic fields (PEMF) have been demonstrated to have anti-inflammatory and pro-regenerative effects in animals and humans. We used the FDA-approved Sofpulse (Ivivi Health Sciences, LLC) to study effect of PEMF on infarct size and poststroke inflammation following distal middle cerebral artery occlusion (dMCAO) in mice. Electromagnetic field was applied within 30-45 min after ischemic brain damage and utilized twice a day for 21 consecutive days. Ischemic infarct size was assessed using MRI and histological analysis. At 21 days after dMCAO, the infarct size was significantly (by 26%) smaller in PEMF-treated animals as compared to controls. Neuroinflammation in these animals was evaluated using specialized cytokine/chemokine PCR array. We demonstrate that PEMF significantly influenced expression profile of pro- and anti-inflammatory factors in the hemisphere ipsilateral to ischemic damage. Importantly, expression of gene encoding major pro-inflammatory cytokine IL-1α was significantly reduced, while expression of major anti-inflammatory IL-10 was significantly increased. PEMF application significantly downregulated genes encoding members of the major pro-apoptotic tumor necrosis factor (TNF) superfamily indicating that the treatment could have both anti-inflammatory and anti-apoptotic effects. Both reduction of infarct size and influence on neuroinflammation could have a potentially important positive impact on the poststroke recovery process, implicating PEMF as a possible adjunctive therapy for stroke patients.
Jonathan Rasouli, Rukmani Lekhraj, Nicholas M White, Eugene S Flamm, Arthur A Pilla, Berish Strauch, Diana Casper
Published 2012 in Neurosciences Letters – DOI: 10.1016/j.neulet.2012.03.089
Abstract
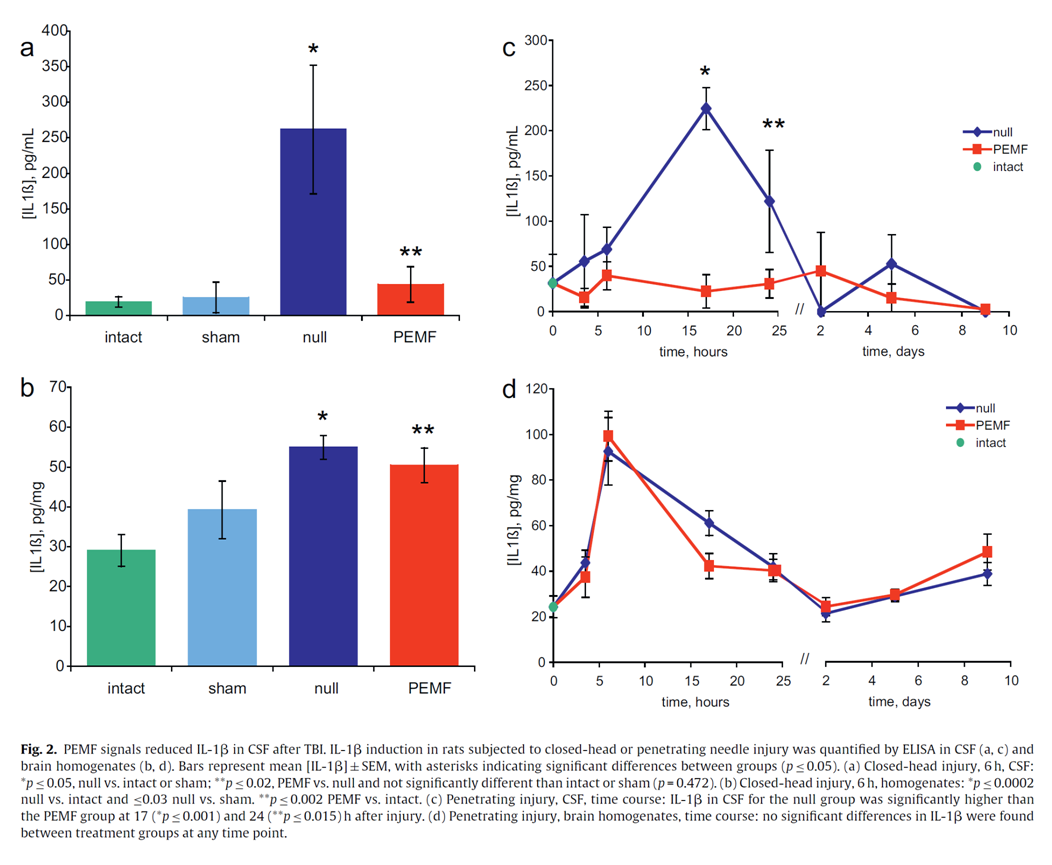
Traumatic Brain Injury (TBI) is a major cause of morbidity and mortality in civilian and military populations. Interleukin-1beta (IL-1β) is a pro-inflammatory cytokine with a key role in the inflammatory response following TBI and studies indicate that attenuation of this cytokine improves behavioral outcomes. Pulsed electromagnetic fields (PEMF) can reduce inflammation after soft tissue injuries in animals and humans. Therefore, we explored whether PEMF signals could alter the course of IL-1β production in rats subjected to closed-head contusive weight-drop injuries (Marmarou method) and penetrating needle-stick brain injuries. Protein levels, measured by the Biorad assay, were not altered by injuries or PEMF treatment. In addition, we verified that IL-1β levels in cerebrospinal fluid (CSF) were proportional to injury severity in the contusion model. Results demonstrate that PEMF treatment attenuated IL-1β levels up to 10-fold in CSF within 6h after contusive injury and also significantly suppressed IL-1β within 17-24h after penetrating injury. In contrast, no differences in IL-1β were seen between PEMF-treated and control groups in brain homogenates. To the authors’ knowledge, this is the first report of the use of PEMF to modulate an inflammatory cytokine after TBI. These results warrant further studies to assess the effects of PEMF on other inflammatory markers and functional outcomes.
D. Bragin, O. Bragina, G. Statom, S. Hagberg, E. Nemoto (University of New Mexico School of Medicine, Neurosurgery, Albuquerque, United States)
Published 2017 in Clinical Neurophysiology – DOI: 10.1016/j.clinph.2016.10.416
Introduction
We previously showed that pulsed electromagnetic field (PEMF) increased microvascular blood flow and tissue oxygenation by nitric oxide-induced vasodilation in a healthy rat brain (Bragin et al., 2015). We also showed that high intracranial pressure (ICP) in rats caused a transition from capillary (CAP) to non-nutritive microvascular shunt (MVS) flow, tissue hypoxia and blood brain barrier (BBB) degradation (Bragin et al., 2011).
Objectives
To evaluate whether PEMF attenuates the detrimental effects of non-nutritive MVS flow induced by high ICP.
Materials and methods
By in vivo 2-photon laser scanning microscopy over the rat parietal cortex, we evaluated the effects of PEMF on microvascular blood flow, tissue oxygenation (NADH), BBB permeability (dye extravasation) and neuronal necrosis (i.v. propidium Iodide) during 4 h of high ICP. Doppler cortical flux, rectal and cranial temperatures, ICP and arterial pressure, blood gases and electrolytes were monitored. After baseline imaging at normal ICP (10 mmHg), rats were subjected to high ICP (30 mmHg) by raising an artificial cerebrospinal fluid reservoir connected to the cisterna magna. At ICP of 30 mmHg, PEMF was applied for 30 min and imaging continuously performed after the treatment. Controls were untreated with PEMF.
Results
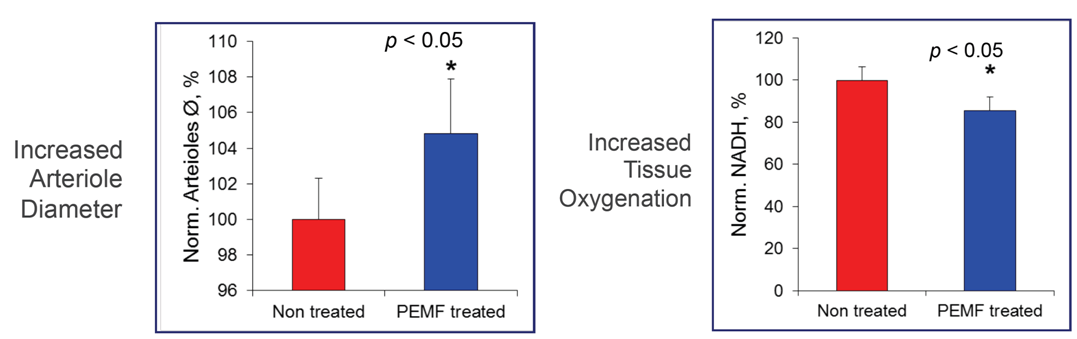
PEMF decreased tissue hypoxia (NADH reduced by 14.6 ± 3.7% compared to control, n = 10 rats per group, mean ± SEM, p < 0.05). BBB damage progression was reduced as reflected by less by 17.2 ± 5.4% dye extravasation (p < 0.05). Decreased by PEMF hypoxia reduced neuronal necrosis (15 ± 3.6% in PEMF vs. 26 ± 6.2% in control, p < 0.05), consistent with dilation of arterioles (+4.5 ± 3.2%) and an increase in capillary blood flow velocity (+4.7 ± 3.2%). PEMF did not completely mitigate the gradual increase in MVS flow at but, as reflected by MVS/capillary ratio, the transition to non-nutritive flow was reduced in the PEMF compared to the untreated rats (2.3 ± 1.1 and 3.8 ± 2.1% change per hour, respectively, p < 0.05).
Conclusions
PEMF reduced tissue hypoxia, BBB degradation and neuronal necrosis by modulating cerebral blood flow at high ICP. PEMF could be an effective treatment for high ICP after severe cerebral insults.
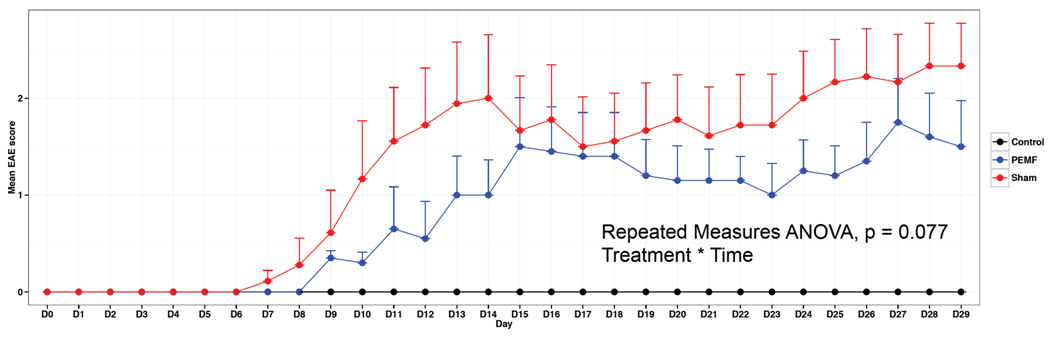
Experimental Autoimmune Encephalomyelitis
Principal Investigator: Sergio Baranzini – University of California, San Francisco
EAE: Inflammatory autoimmune model of multiple sclerosis
Scoring: 0 (healthy) – 5 (moribund)
PEMF or Sham treatment of mice with EAE (n = 10 per group) for 15 minutes twice per day
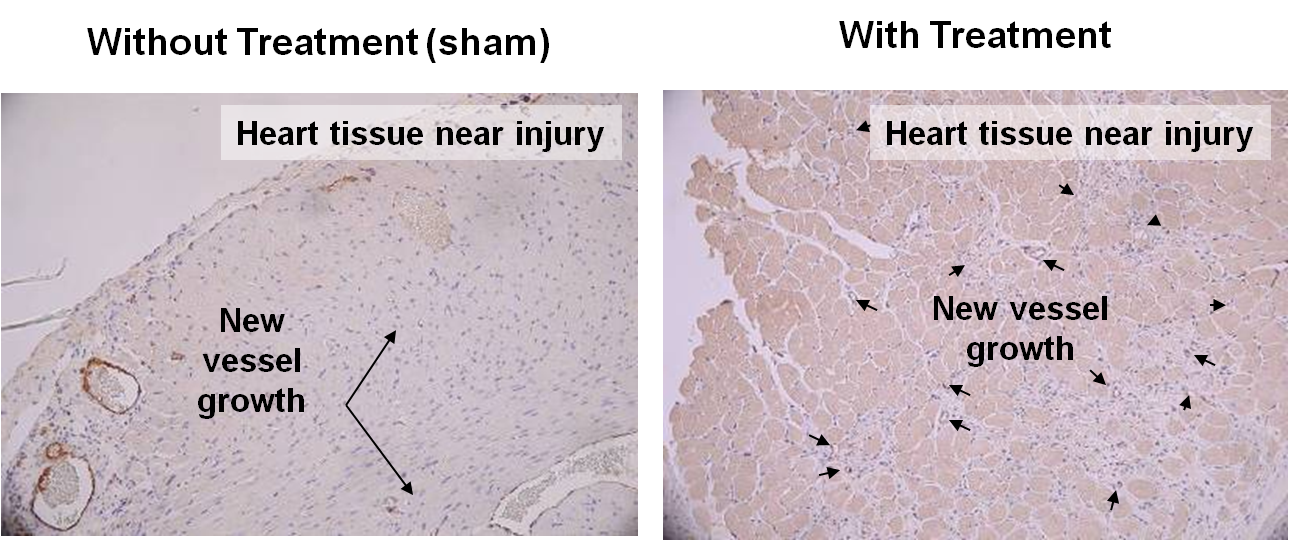
Pulsed Electromagnetic Fields (PEMF) Effects on Injured Heart in Animal Model
100% Increase in New Blood Vessels in Treated Animals